Synthesis and Photocatalytic Performance Evaluation of BaTiO3/ZnO/Cs3Bi2I9 Based Perovskite for Solar Cell Applications
DOI:
https://doi.org/10.22452/mjs.vol44no3.5Keywords:
Perovskites, Solar cells, Nanoparticles, Thermal method, Barium titanateAbstract
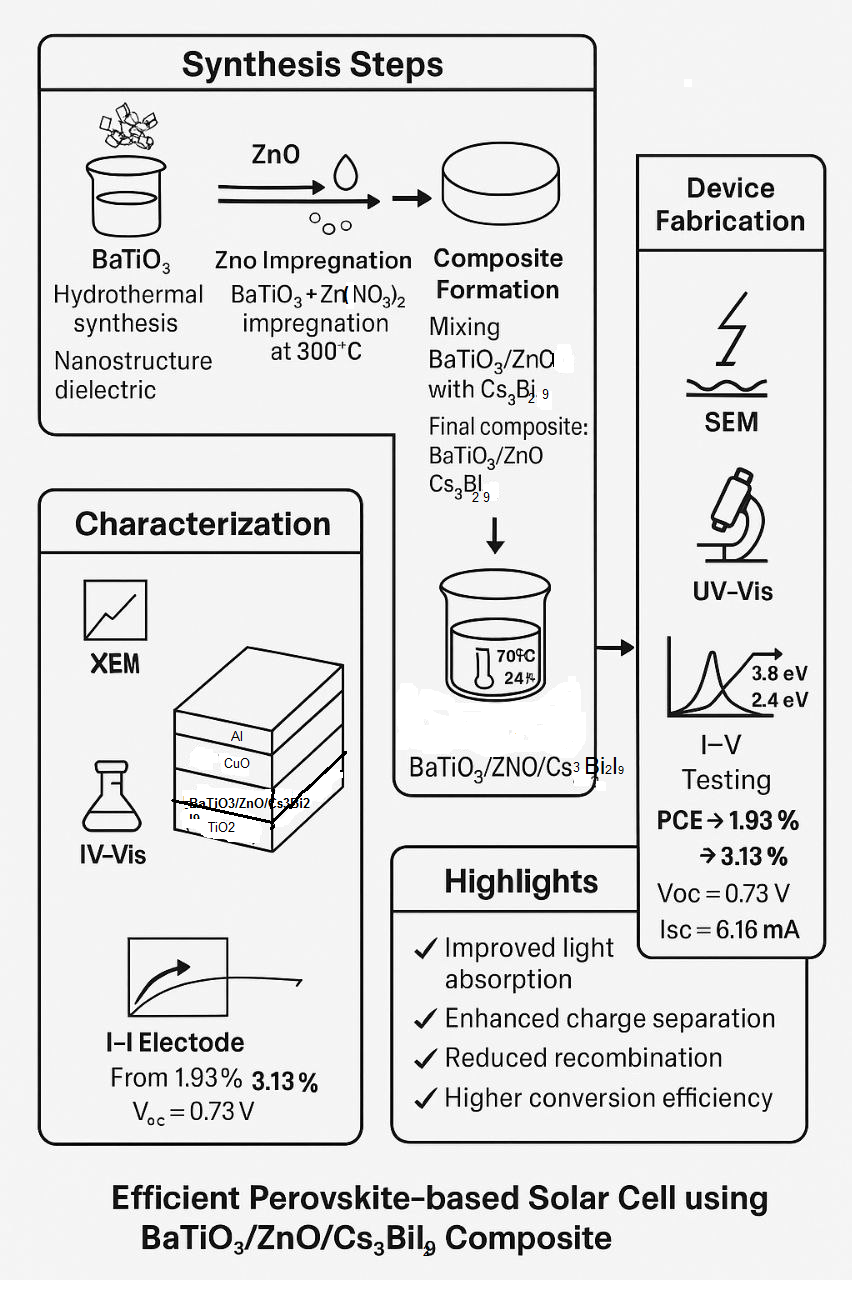
Due to the light absorption properties of perovskites, including halides, perovskite cells are considered an ideal energy system. This study aims to improve photoexcitation separation by introducing a material with a perovskite-like structure, such as Cs3Bi2I9, alone or with barium titanate nanoparticles as a second choice, while the third choice is to impregnate barium titanate (prepared by hydrothermal method) with ZnO to form BaTiO3/ZnO, then mix it with Cs3Bi2I9. All these choices are fabricated as a sandwich between the n-type and p-type collection. The produced layers, BaTiO3/ZnO /Cs3Bi2I9, were characterized using XRD, EDX, SEM, and UV-Vis spectroscopic analytical techniques. The results suggest that the band gap of the prepared layer was further decreased compared to the original material, Cs3Bi2I9. The performance test revealed a photo conversion efficiency (PCE) of 3.13% and a highest power of 3.15 MW, comparable to or higher than other studies. This suggests that this layer significantly reduces recombination phenomena and improves the cell's performance overall.
References
Abd, A. N. (2023). Free-Lead Perovskite Materials , CsFeCl3 And Cs3Fe2Cl9 , In Solar Cell. ACE Journal of Advance Research In Physical Sciences, July. https://doi.org/10.59218/makacejarps.2023.12.19
Al-Marzouki, F. M., Al-Hartomy, O. A., & Shah, M. A. (2011). Preparation of copper oxide (CuO) nanoparticles and their bactericidal activity. International Journal of Manufacturing, Materials, and Mechanical Engineering, 1(4), 58–64. https://doi.org/10.4018/ijmmme.2011100104
Arjmand, F., Fatemi, S. J., Maghsoudi, S., & Naeimi, A. (2022). The first and cost effectivenano-biocomposite, zinc porphyrin/ CuO/reduced graphene oxide, based on Calotropis procera plant for perovskite solar cellas hole-transport layerunderambient conditions. Journal of Materials Research and Technology, 16, 1008–1020. https://doi.org/10.1016/j.jmrt.2021.12.012
Article, R. (2016). A Brief Review on Synthesis and Characterization of Copper Oxide Nanoparticles and its Applications. Journal of Bioelectronics and Nanotechnology, 1(1), 1–9. https://doi.org/10.13188/2475-224x.1000003
Bai, F., Hu, Y., Hu, Y., Qiu, T., Miao, X., & Zhang, S. (2018). he first and cost effectivenano-biocomposite, zinc porphyrin/ CuO/reduced graphene oxide, based on Calotropis procera plant for perovskite solar cellas hole. Solar Energy Materials and Solar Cells, 184(January), 15–21. https://doi.org/10.1016/j.solmat.2018.04.032
Basaleh, A. S., & Mohamed, R. M. (2020). Synthesis and characterization of Cu-BaTiO3 nanocomposite for atrazine remediation under visible-light radiation from wastewater. Journal of Materials Research and Technology, 9(5), 9550–9558. https://doi.org/10.1016/j.jmrt.2020.06.081
Dorris, S. E., & Kumarakrishnan, S. (1983). Zyxw Zyxwvuts. 94(December).
Gunerhan, H., Hepbasli, A., & Giresunlu, U. (2009). Environmental impacts from the solar energy systems. Energy Sources, Part A: Recovery, Utilization and Environmental Effects, 31(2), 1131–1138. https://doi.org/10.1080/15567030701512733
Hamed, T. A., & Alshare, A. (2022). Environmental Impact of Solar and Wind energy-A Review. Journal of Sustainable Development of Energy, Water and Environment Systems, 10(2), 1–23. https://doi.org/10.13044/j.sdewes.d9.0387
Harbbi, K. H., & Jahil, S. S. (2017). Study the Lattice Distortion and Particle Size of One Phase of MnO by Using Fourier Analysis of X-ray Diffraction Lines. Advances in Physics Theories and Applications, 65(x), 6–22.
Jassim, G., Najim, M., & Salih, W. (2021). Preparation of Micro Barium Titanate Powder and Comparison with Nano Powder Properties. Journal of Applied Sciences and Nanotechnology, 1(4), 12–23. https://doi.org/10.53293/jasn.2021.3653.1033
Maison, W., Kleeberg, R., Heimann, R. B., & Phanichphant, S. (2003). Phase content, tetragonality, and crystallite size of nanoscaled barium titanate synthesized by the catecholate process: Effect of calcination temperature. Journal of the European Ceramic Society, 23(1), 127–132. https://doi.org/10.1016/S0955-2219(02)00071-7
Mehta, S. K., Kumar, S., Chaudhary, S., & Bhasin, K. K. (2009). Supplementary Material (ESI) for Nanoscale Supplementary data Nucleation and growth of surfactant passivated CdS and HgS NPs: Time dependent Absorption and Luminescence profiles. The Royal Society of Chemistry, c, 1–6.
Omar, A., & Abdullah, H. (2014). Electron transport analysis in zinc oxide-based dye-sensitized solar cells: A review. Renewable and Sustainable Energy Reviews, 31, 149–157. https://doi.org/10.1016/j.rser.2013.11.031
Park, B. W., Philippe, B., Zhang, X., Rensmo, H., Boschloo, G., & Johansson, E. M. J. (2015). B. W. Park, B. Philippe. Advanced Materials, 27(43), 6806–6813. https://doi.org/10.1002/adma.201501978
Pescoe, ©, & Ali, F. M. (n.d.). A Review on Solar Energy Potential And Future world.
Rabiei, M., Palevicius, A., Monshi, A., Nasiri, S., Vilkauskas, A., & Janusas, G. (2020). Comparing methods for calculating nano crystal size of natural hydroxyapatite using X-ray diffraction. Nanomaterials, 10(9), 1–21. https://doi.org/10.3390/nano10091627
Ray, A. K. (2007). Synthesis and Charactrization Of BaTiO3 Powder Prepared By Combustion Synthesis Process
Sengupta, L. C., Stowell, S., Ngo, E., Oday, M. E., & Lancto, R. (1995). Barium strontium titanate and nonferroelectric oxide ceramic composites for use in phased array antennas. Integrated Ferroelectrics, 8(1–2), 77–88. https://doi.org/10.1080/10584589508012302
Tanaka, H., Oku, T., & Ueoka, N. (2018). Structural stabilities of organic–inorganic perovskite crystals. Japanese Journal of Applied Physics, 57(8), 0–9. https://doi.org/10.7567/JJAP.57.08RE12
Tanaka, K., Takahashi, T., Ban, T., Kondo, T., Uchida, K., & Miura, N. (2003). Comparative study on the excitons in lead-halide-based perovskite-type crystals CH3NH3PbBr3 CH3NH 3PbI3. Solid State Communications, 127(9–10), 619–623. https://doi.org/10.1016/S0038-1098(03)00566-0
Tomaszewski, P. E. (1994). Crystal Structure and Phase Transitions in the A3B2X9 Family of Crystals. Physica Status Solidi (B), 181(1), 15–21. https://doi.org/10.1002/pssb.2221810102
viewcontent.cgi. (n.d.).
Wang, M., Wang, W., Ma, B., Shen, W., Liu, L., Cao, K., Chen, S., & Huang, W. (2021). Lead-Free Perovskite Materials for Solar Cells. In Nano-Micro Letters (Vol. 13, Issue 1). Springer Singapore. https://doi.org/10.1007/s40820-020-00578-z
Zhang, C., Li, X., Ding, L., Jin, C., & Tao, H. (2022). Effect of BaTiO3 powder as an additive in perovskite films on solar cells. RSC Advances, 12(13), 7950–7960. https://doi.org/10.1039/d1ra09374f
Zhang, Z., Li, X., Xia, X., Wang, Z., Huang, Z., Lei, B., & Gao, Y. (2017). High-Quality (CH3NH3)3Bi2I9 Film-Based Solar Cells: Pushing Efficiency up to 1.64%. Journal of Physical Chemistry Letters, 8(17), 4300–4307. https://doi.org/10.1021/acs.jpclett.7b01952
zou2012.pdf. (n.d.).
Downloads
Published
Issue
Section
License
Copyright (c) 2025 Malaysian Journal of Science

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
Transfer of Copyrights
- In the event of publication of the manuscript entitled [INSERT MANUSCRIPT TITLE AND REF NO.] in the Malaysian Journal of Science, I hereby transfer copyrights of the manuscript title, abstract and contents to the Malaysian Journal of Science and the Faculty of Science, University of Malaya (as the publisher) for the full legal term of copyright and any renewals thereof throughout the world in any format, and any media for communication.
Conditions of Publication
- I hereby state that this manuscript to be published is an original work, unpublished in any form prior and I have obtained the necessary permission for the reproduction (or am the owner) of any images, illustrations, tables, charts, figures, maps, photographs and other visual materials of whom the copyrights is owned by a third party.
- This manuscript contains no statements that are contradictory to the relevant local and international laws or that infringes on the rights of others.
- I agree to indemnify the Malaysian Journal of Science and the Faculty of Science, University of Malaya (as the publisher) in the event of any claims that arise in regards to the above conditions and assume full liability on the published manuscript.
Reviewer’s Responsibilities
- Reviewers must treat the manuscripts received for reviewing process as confidential. It must not be shown or discussed with others without the authorization from the editor of MJS.
- Reviewers assigned must not have conflicts of interest with respect to the original work, the authors of the article or the research funding.
- Reviewers should judge or evaluate the manuscripts objective as possible. The feedback from the reviewers should be express clearly with supporting arguments.
- If the assigned reviewer considers themselves not able to complete the review of the manuscript, they must communicate with the editor, so that the manuscript could be sent to another suitable reviewer.
Copyright: Rights of the Author(s)
- Effective 2007, it will become the policy of the Malaysian Journal of Science (published by the Faculty of Science, University of Malaya) to obtain copyrights of all manuscripts published. This is to facilitate:
- Protection against copyright infringement of the manuscript through copyright breaches or piracy.
- Timely handling of reproduction requests from authorized third parties that are addressed directly to the Faculty of Science, University of Malaya.
- As the author, you may publish the fore-mentioned manuscript, whole or any part thereof, provided acknowledgement regarding copyright notice and reference to first publication in the Malaysian Journal of Science and Faculty of Science, University of Malaya (as the publishers) are given. You may produce copies of your manuscript, whole or any part thereof, for teaching purposes or to be provided, on individual basis, to fellow researchers.
- You may include the fore-mentioned manuscript, whole or any part thereof, electronically on a secure network at your affiliated institution, provided acknowledgement regarding copyright notice and reference to first publication in the Malaysian Journal of Science and Faculty of Science, University of Malaya (as the publishers) are given.
- You may include the fore-mentioned manuscript, whole or any part thereof, on the World Wide Web, provided acknowledgement regarding copyright notice and reference to first publication in the Malaysian Journal of Science and Faculty of Science, University of Malaya (as the publishers) are given.
- In the event that your manuscript, whole or any part thereof, has been requested to be reproduced, for any purpose or in any form approved by the Malaysian Journal of Science and Faculty of Science, University of Malaya (as the publishers), you will be informed. It is requested that any changes to your contact details (especially e-mail addresses) are made known.
Copyright: Role and responsibility of the Author(s)
- In the event of the manuscript to be published in the Malaysian Journal of Science contains materials copyrighted to others prior, it is the responsibility of current author(s) to obtain written permission from the copyright owner or owners.
- This written permission should be submitted with the proof-copy of the manuscript to be published in the Malaysian Journal of Science
Licensing Policy
Malaysian Journal of Science is an open-access journal that follows the Creative Commons Attribution-Non-commercial 4.0 International License (CC BY-NC 4.0)
CC BY – NC 4.0: Under this licence, the reusers to distribute, remix, alter, and build upon the content in any media or format for non-commercial purposes only, as long as proper acknowledgement is given to the authors of the original work. Please take the time to read the whole licence agreement (https://creativecommons.org/licenses/by-nc/4.0/legalcode ).





