Safety Assessment of One Day Treatment of Liquorice Extract in Female Sprague Dawley Rats
DOI:
https://doi.org/10.22452/mjs.vol44sp1.9Keywords:
Liquorice extract, hepatic function, renal function, serum biochemical markerAbstract
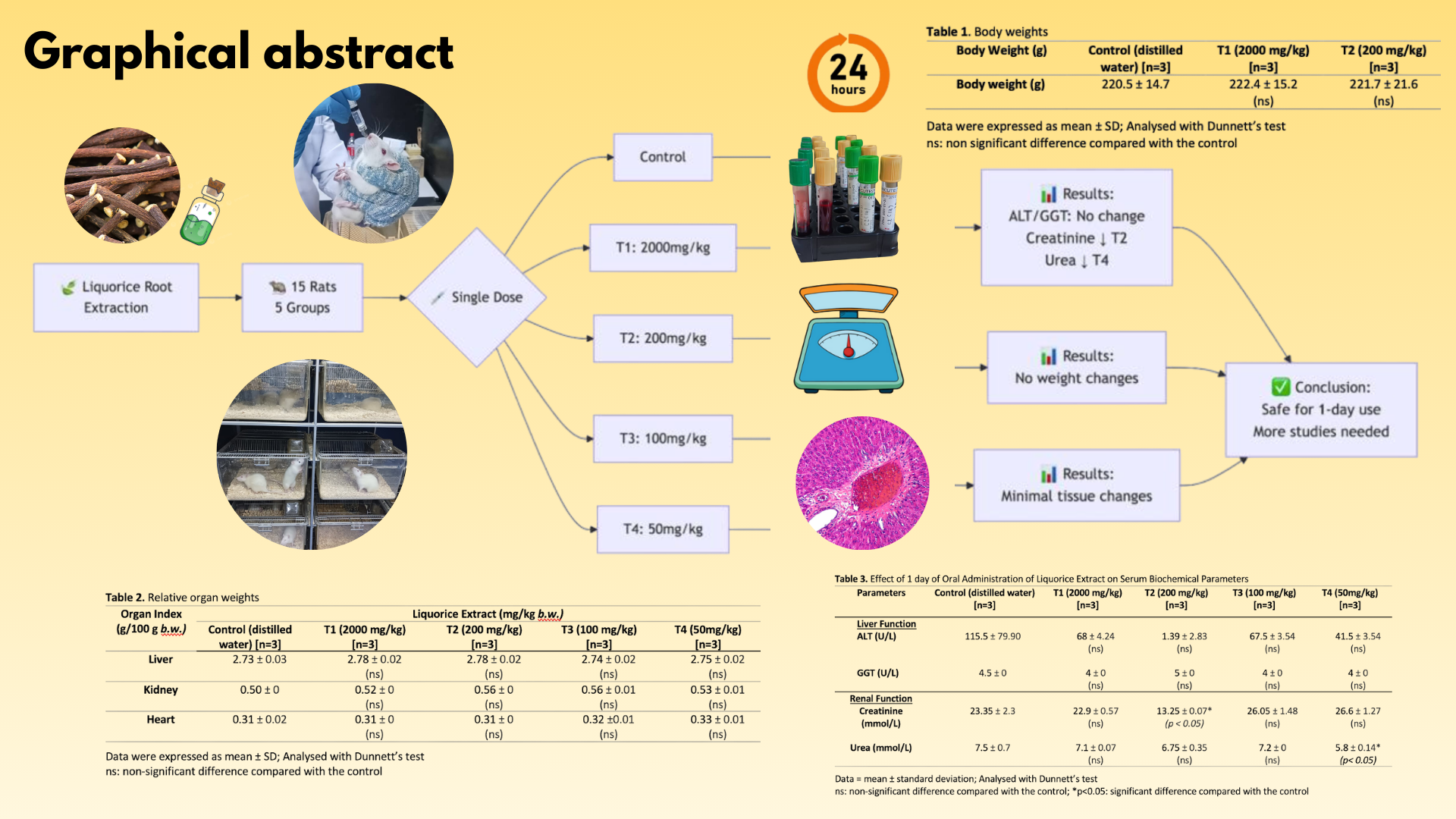
This study aimed to provide preliminary data on the safe use of liquorice extracts for herbal product consumers. Female Sprague Dawley rats received a single-day treatment with liquorice extract (50–2000 mg/kg) or distilled water (control). Liver and kidney functions were evaluated through blood biochemical analysis, gross, and histological evaluation. This animal study adhered to the OECD Test Guideline 423. Fifteen female SD rats, aged 16 weeks, were randomly assigned to five groups (n = 3). The control group received distilled water, while treatment groups T1, T2, T3, and T4 were administered liquorice extract at doses of 2000, 200, 100, and 50 mg/kg via oral gavage. Body and organ weights (liver, kidney, heart) were measured. Blood samples were collected to assess serum biochemical markers, including alanine aminotransferase (ALT), gamma-glutamyl transferase (GGT), creatinine, and urea levels. Data were analysed using Dunnett’s tests, with p < 0.05 denoting statistical significance. No significant changes in body or organ weights occurred across groups. Liquorice extract had no effect on ALT or GGT. Serum creatinine decreased (p < 0.05) at 200 mg/kg, and serum urea decreased at 50 mg/kg compared with controls. A single-day liquorice extract treatment (50–2000 mg/kg) was safe and caused no hepatic or renal toxicity in female SD rats.
References
Abdel Maksoud, H. A., Abdel Magid, A. D., Mostafa, Y. M., Elharrif, M. G., Sorour, R. I., & Sorour, M. I. (2019). Ameliorative effect of liquorice extract versus silymarin in experimentally induced chronic hepatitis: A biochemical and genetic study. Clinical Nutrition Experimental, 23, 69–79.
Assar, D. H., Elhabashi, N., Mokhbatly, A. A. A., Ragab, A. E., Elbialy, Z. I., Rizk, S. A., et al. (2021). Wound healing potential of licorice extract in rat model: Antioxidants, histopathological, immunohistochemical and gene expression evidences. Biomedicine & Pharmacotherapy, 143, 112-151
Brookes, E. M., & Power, D. A. (2022). Elevated serum urea-to-creatinine ratio is associated with adverse inpatient clinical outcomes in non-end stage chronic kidney disease. Scientific Reports, 12(1), 20827. Retrieved from https://www.nature.com/articles/s41598-022-25254-7
Dang, L., Jin, Y., Yuan, Y., Shao, R., & Wang, Y. (2024). The King of Chinese Medicine—Glycyrrhiza glabra (Licorice): All-round Inquiry in its Chemical Composition, Pharmacodynamics, Traditional and Medicinal Value. Acupuncture and Herbal Medicine. https://doi.org/10.1097/hm9.0000000000000103
GGT – eClinpath. (n.d.). eClinpath.com. Retrieved from https://eclinpath.com/chemistry/liver/cholestasis/gamma-glutamyl-transferase/
Gomaa, A. A., & Abdel-Wadood, Y. A. (2021). The potential of glycyrrhizin and licorice extract in combating COVID-19 and associated conditions. Phytomedicine Plus, 1(3), 100043. https://doi.org/10.1016/j.phyplu.2021.100043
Gounden, V., & Jialal, I. (2024). Renal function tests. National Library of Medicine, StatPearls Publishing. Retrieved from https://www.ncbi.nlm.nih.gov/books/NBK507821/
Husain, I., Bala, K., Khan, I. A., & Khan, S. I. (2021). A review on phytochemicals, pharmacological activities, drug interactions, and associated toxicities of licorice (Glycyrrhiza sp.). Food Frontiers, 2(4), 449-485. https://doi.org/10.1002/fft2.110
Kim, H. Y., Zuo, G., Lee, S. K., & Lim, S. S. (2020). Acute and subchronic toxicity study of nonpolar extract of licorice roots in mice. Food Science & Nutrition, 8(5), 2242–2250. https://doi.org/10.1002/fsn3.1465
Lajoie, L., Fabiano-Tixier, A. S., & Chemat, F. (2022). Water as a green solvent: Methods of solubilization and extraction of natural products—Past, present and future solutions. Pharmaceuticals, 15(12), 1507
Lazic, S. E., Semenova, E., & Williams, D. P. (2020). Determining organ weight toxicity with Bayesian causal models: Improving on the analysis of relative organ weights. Scientific Reports, 10(1).
Moriles, K. E., & Azer, S. A. (2020). Alanine amino transferase. National Library of Medicine, StatPearls Publishing. Retrieved from https://www.ncbi.nlm.nih.gov/books/NBK559278/
Murray, M. T. (2020). Glycyrrhiza glabra (licorice). In Textbook of Natural Medicine (3rd ed., pp. 641–647). https://doi.org/10.1016/B978-0-323-43044-9.00085-6
Pawar, B., Gupta, T., Vasdev, N., Tekade, M., Arafat, B., & Tekade, R. K. (2023). Understanding pharmacotoxicology. In R. K. Tekade (Ed.), Basic fundamentals of drug delivery (pp. 1–28). Elsevier. https://doi.org/10.1016/b978-0-443-15840-7.00025-7
Plaskova, A., & Mlcek, J. (2023). New insights of the application of water or ethanol-water plant extract rich in active compounds in food. Frontiers in Nutrition, 10, 1118761. Retrieved from https://pubmed.ncbi.nlm.nih.gov/37057062/
Sharifi-Rad, J., Quispe, C., Herrera-Bravo, J., Herrera Belén, L., Kaur, R., Kregiel, D., et al. (2021). Glycyrrhiza genus: Enlightening phytochemical components for pharmacological and health-promoting abilities. Oxidative Medicine and Cellular Longevity, 2021, 7571132. https://doi.org/10.1155/2021/7571132
Sun, B., Kelleher, S., Short, C., Valencia, P. A., & Zagory, J. A. (2021). Recent advancements in laboratory screening, diagnosis, and prognosis of biliary atresia: A literature review. Digestive Medicine Research, 4(0). Retrieved from https://dmr.amegroups.org/article/view/7592/html
Thakur, S., Kumar, V., Das, R., Sharma, V., & Mehta, D. K. (2024). Biomarkers of hepatic toxicity: An overview. Current Therapeutic Research, 100, 100737. https://doi.org/10.1016/j.curtheres.2024.100737
Wahab, S., Annadurai, S., Abullais, S. S., Das, G., Ahmad, W., Ahmad, M. F., Kandasamy, G., Vasudevan, R., Ali, M. S., & Amir, M. (2021). Glycyrrhiza glabra (Licorice): A comprehensive review on its phytochemistry, biological activities, clinical evidence and toxicology. (n.d.). Plants, 10(12), 275. https://doi.org/10.3390/plants1012275
Wang, Z., Ma, J., Yao, S., He, Y., Miu, K. K., Xia, Q., et al. (2022). Liquorice extract and 18β-glycyrrhetinic acid protect against experimental pyrrolizidine alkaloid-induced hepatotoxicity in rats through inhibiting cytochrome P450-mediated metabolic activation. Frontiers in Pharmacology, 13, 850-859. https://doi.org/10.3389/fphar.2022.850859
Downloads
Published
Issue
Section
License
Copyright (c) 2025 Malaysian Journal of Science

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
Transfer of Copyrights
- In the event of publication of the manuscript entitled [INSERT MANUSCRIPT TITLE AND REF NO.] in the Malaysian Journal of Science, I hereby transfer copyrights of the manuscript title, abstract and contents to the Malaysian Journal of Science and the Faculty of Science, University of Malaya (as the publisher) for the full legal term of copyright and any renewals thereof throughout the world in any format, and any media for communication.
Conditions of Publication
- I hereby state that this manuscript to be published is an original work, unpublished in any form prior and I have obtained the necessary permission for the reproduction (or am the owner) of any images, illustrations, tables, charts, figures, maps, photographs and other visual materials of whom the copyrights is owned by a third party.
- This manuscript contains no statements that are contradictory to the relevant local and international laws or that infringes on the rights of others.
- I agree to indemnify the Malaysian Journal of Science and the Faculty of Science, University of Malaya (as the publisher) in the event of any claims that arise in regards to the above conditions and assume full liability on the published manuscript.
Reviewer’s Responsibilities
- Reviewers must treat the manuscripts received for reviewing process as confidential. It must not be shown or discussed with others without the authorization from the editor of MJS.
- Reviewers assigned must not have conflicts of interest with respect to the original work, the authors of the article or the research funding.
- Reviewers should judge or evaluate the manuscripts objective as possible. The feedback from the reviewers should be express clearly with supporting arguments.
- If the assigned reviewer considers themselves not able to complete the review of the manuscript, they must communicate with the editor, so that the manuscript could be sent to another suitable reviewer.
Copyright: Rights of the Author(s)
- Effective 2007, it will become the policy of the Malaysian Journal of Science (published by the Faculty of Science, University of Malaya) to obtain copyrights of all manuscripts published. This is to facilitate:
- Protection against copyright infringement of the manuscript through copyright breaches or piracy.
- Timely handling of reproduction requests from authorized third parties that are addressed directly to the Faculty of Science, University of Malaya.
- As the author, you may publish the fore-mentioned manuscript, whole or any part thereof, provided acknowledgement regarding copyright notice and reference to first publication in the Malaysian Journal of Science and Faculty of Science, University of Malaya (as the publishers) are given. You may produce copies of your manuscript, whole or any part thereof, for teaching purposes or to be provided, on individual basis, to fellow researchers.
- You may include the fore-mentioned manuscript, whole or any part thereof, electronically on a secure network at your affiliated institution, provided acknowledgement regarding copyright notice and reference to first publication in the Malaysian Journal of Science and Faculty of Science, University of Malaya (as the publishers) are given.
- You may include the fore-mentioned manuscript, whole or any part thereof, on the World Wide Web, provided acknowledgement regarding copyright notice and reference to first publication in the Malaysian Journal of Science and Faculty of Science, University of Malaya (as the publishers) are given.
- In the event that your manuscript, whole or any part thereof, has been requested to be reproduced, for any purpose or in any form approved by the Malaysian Journal of Science and Faculty of Science, University of Malaya (as the publishers), you will be informed. It is requested that any changes to your contact details (especially e-mail addresses) are made known.
Copyright: Role and responsibility of the Author(s)
- In the event of the manuscript to be published in the Malaysian Journal of Science contains materials copyrighted to others prior, it is the responsibility of current author(s) to obtain written permission from the copyright owner or owners.
- This written permission should be submitted with the proof-copy of the manuscript to be published in the Malaysian Journal of Science
Licensing Policy
Malaysian Journal of Science is an open-access journal that follows the Creative Commons Attribution-Non-commercial 4.0 International License (CC BY-NC 4.0)
CC BY – NC 4.0: Under this licence, the reusers to distribute, remix, alter, and build upon the content in any media or format for non-commercial purposes only, as long as proper acknowledgement is given to the authors of the original work. Please take the time to read the whole licence agreement (https://creativecommons.org/licenses/by-nc/4.0/legalcode ).





